ORIGINAL RESEARCH REPORTS
VoluDerm microneedle technology for skin treatments—In vivo histological evidencel
Abstract
Introduction: The growing demand for skin rejuvenation treatments and procedures with minimal down time and low risk has led to the development of fractional radiofrequency (RF) systems. The new VoluDerm™ RF microneedle technology creates minute columns of tissue thermal microablation. Treatment triggers natural fractional healing, resulting in dermal volumizing and skin renewal. This preclinical research assessed the safety and efficacy of the VoluDerm microneedling device through histological evaluation of morphological changes in the target tissue. Methods: Following approval of protocol by ethical committee, treatments were conducted on two domestic pigs using VoluDerm disposable tips. Histological samples of 14, 7, 4 days and immediately after treatment with various energy settings were analyzed. Results: Immediate VoluDerm epidermal and dermal effects, and progress of healing process, as function of time following treatment (days 4 and 7), were demonstrated. Histology analysis of samples of 14 days demonstrated complete healing for all energy levels. Summary: This in vivo histology con-firmed the safe and effective performance of the VoluDerm treatment. A fractional pattern of affected areas, surrounded by healthy tissue, was demonstrated. Healing process proved natural dermal renewal and epidermal complete regeneration. Histology supports clinical advantages of the VoluDerm natural-looking skin enhancement, with none to minimal pain and no downtime.
Key Words: antiaging, fractional, histology, radiofrequency, VoluDerm
See also: Tripollar Pose VX Skin Renewal Therapy, Tripollar Prism Led Skin Mask, Tripollar Stop VX2 Home Facial Device
Introduction
Non-surgical medical aesthetic treatments and treatments for skin rejuvenation, aiming to improve the appearance of facial wrinkles, reduce acne scars with treatments, and improve skin imperfections, range from non-ablative, minimal ablative, to ablative procedures. Non-ablative lasers, intense pulsed light (IPL), and radiofrequency (RF) technologies, especially the apollo® (Pollogen Ltd.) and Maximus® (Pollogen Ltd.) systems based on the TriPollar® RF technology, are widely used for various skin treatment indications via a thermal volumetric heating of the target tissue, leading to dermal collagen remodeling (1–8). These non-ablative procedures are considered as having a high safety profile and moderate clinical effect. On the other hand, ablative resurfacing lasers, such as carbon dioxide (CO2) lasers and Erbium:Yttrium– Aluminum–garnet (Erbium YAG) lasers, were con-sidered the gold standard for minimally invasive treatments. However, these treatments were associ-ated with significant downtime and relatively high risk of side effect (9–12), especially on dark and Asian skin types. The demand for new procedures delivering visible clinical improvement with reduced downtime has led to the development of fractional lasers (13,14). Instead of treating an entire skin area, these lasers treat a condensed matrix of small “islets” of tissue leaving intact skin between these islets. Healing is initiated from these intact skin areas, resulting in reduced downtime and reduced compli-cations. Since the introduction of the first fractional laser system, a myriad of “fractional” laser systems, mostly based on CO2 and Erbium YAG laser technology, have been developed and approved for marketing worldwide (15).
In the past several years, several fractional RF systems have been introduced that allow controlled fractional skin resurfacing by employing a matrix of miniature RF electrodes placed in contact with the skin surface. In addition to the epidermal effect, frac-tional RF systems allow enhancement of the dermal layer via volumetric heating, thus leading to an effec-tive microablative skin resurfacing and improvement in appearance of wrinkles, fine lines, and acne scars. Clinical and histological results of such fractional RF systems, including the TriFractional™ RF technology from Pollogen Ltd. for microablative skin resurfacing and the treatment of wrinkles, were published (16–18). The fractional treatments mainly lead to con-trolled epidermal microablation along with concomitant dermal remodeling, due to the indirect volumetric heating.
Recently, new devices employing minimally inva-sive bipolar microneedle RF electrodes, which are inserted percutaneously into the skin, were intro-duced into the market for the treatment of wrinkles and skin laxity (19–23). Published studies revealed that such treatments induced an active dermal remod-eling process including neocollagenesis and neoelas-togenesis, with an almost intact epidermal layer. The current research was intended to evaluate the histological dermal and epidermal effects follow-ing treatment with Pollogen VoluDerm microneedle RF technology for skin volume enhancement and wrinkles treatment.
Methods
VoluDerm technology
The VoluDerm technology uses a disposable tip attached to the distal end of the applicator (Figure 1), which is placed on the target skin area. The tip is composed of an array of microneedles (0.5 mm in length), which are inserted into the skin via RF- assisted penetration, enabling an efficient, smooth, uniform, and homogenous penetration with none to minimal pain. Two types of tips are available: gen 36 and gen 12 with 36 and 12 microneedles, respec-tively. RF had frequency of 1 MHZ, power of up to 44 W, and pin electrode energy of up to 62mj/pin. VoluDerm RF energy creates invisible microwounds, characterized by an optimized dermal impact and minimal epidermal effect. The microwounds are sur-rounded by healthy unaffected skin areas, which function as healing centers, promoting neocollagen-esis and skin renewal in a safe and effective process, with minimal pain and side effects.
The VoluDerm treatment triggers the body’s nat-ural healing response involving inflammatory, prolif-erative, and remodeling stages. Blood vessels-related reactions include vasodilatation; increased porosity of blood vessels to facilitate the gradual entry of inflammatory cells like leukocytes and neutrophils, fibronectin, and growth factors into the wound site from the bloodstream; and angiogenesis (24,25). Natural process continues with reepithelialization and remodeling until complete healing of micro-wounds is achieved.
Study procedure
Animal model and study site. Two domestic pigs, weighing 20–25 kg, were used in this study. Study was conducted at GLPigs site, Assaf Harofeh, Israel, following approval of study protocol by the ethics committee.
Animal and skin preparation. Treatment was con-ducted in a complete surgical theater under general anesthesia. Pretreatment: animal skin was shaved at treatment site and the animal was placed in dorsal recumbence. The animal’s back was marked, initially using surgical marker and then using tattoo to form two sets of marked 1.5 3 1.5 cm squares from the spine laterally. Each square represented an applica-tion site corresponding to an index table.
Treatment settings. A single treatment per setting and time point, using gen12 and gen36 disposable tips, was conducted on the back skin of the pig. Each pig was treated on the back area over both sides of the spine. In order to avoid possible lateral influence, the sites were treated alternately.
Three energy level settings were used and each treatment was performed in duplicate so that each setting was performed twice—once on each pig.

Study timeline. Upon study commencement, all sites were marked with tattoo. Four treatments were per-formed; on day 1 (14-day histology sample), on day 7 (7-day histology sample), day 10 (4-day histology sample), and on day 14 (immediate effect—0-day sample). Animals were kept in standard husbandry until the next time point. On the last day, both the animals were sacrificed.
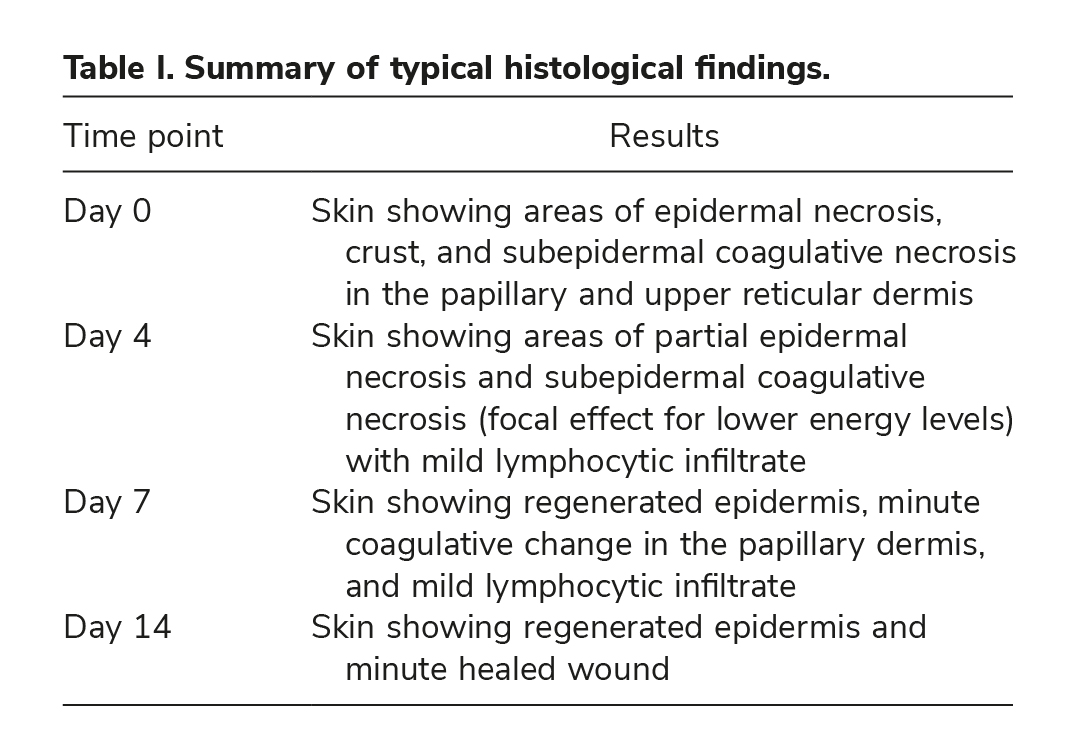
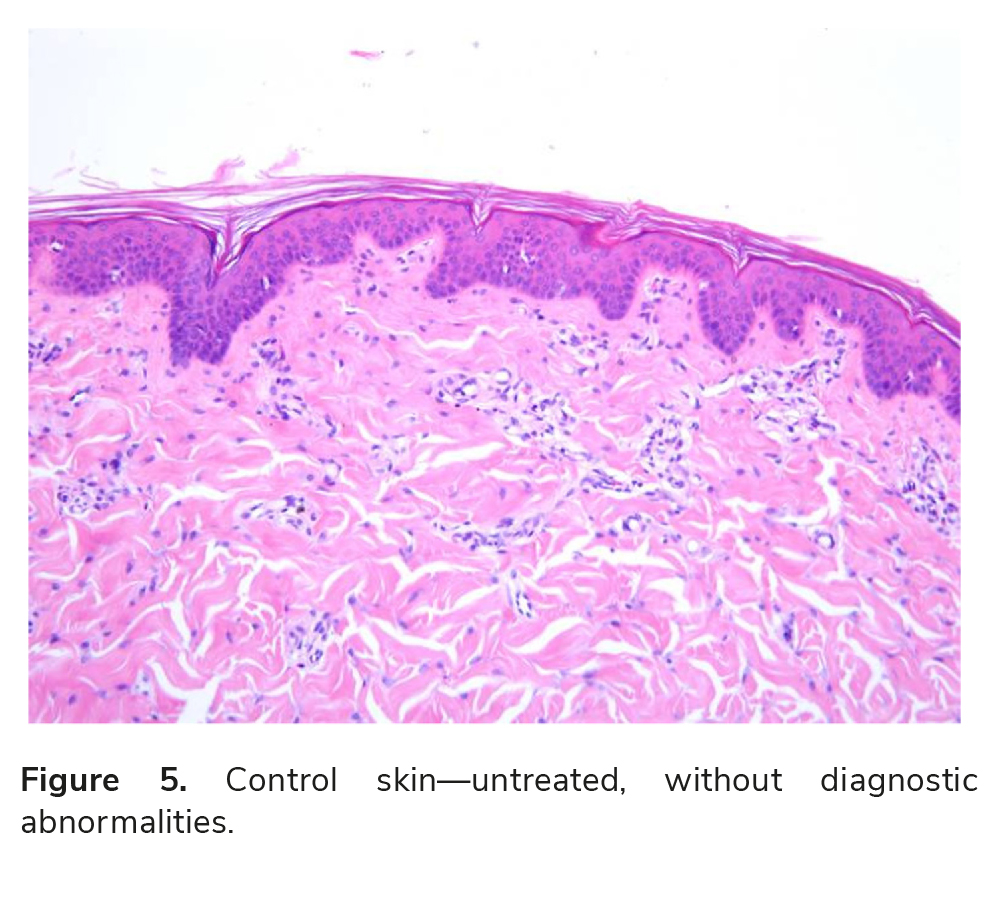
Skin samples. Skin samples for histological and mor-phological analysis were collected for the following time points: immediately after treatment, 4, 7, and 14 days post treatment. In addition, a control skin sample was collected.
Following excision, skin samples were fixed in formalin and embedded in paraffin. Serial sections were prepared and stained using hematoxylin and eosin (H&E) for dermatopathological examination and analysis.
Histological analysis. Histological analysis was per-formed by an Anatomic Pathology specialist. Slides were provided for analysis marked by numbers which do not reflect the slide identification in terms of the time point and energy level used.
Discussion
Hantash et al. (19) were the first to report on a bipolar, microneedle, fractional RF device (Renesis™, Primaeva Medical Inc. Ca.). This system delivers up to 35 W RF energy directly within human dermis via 5 microneedle electrode pairs. Tissue temperature is held at 72o C for 4 seconds using a feedback system. Superficial cooling is achieved using a Peltier cooling device. Treatment was administered to patients prior to their scheduled abdominoplasty at different time intervals. The wound-healing response was evaluated histologically and semiquantitatively using chemical enzymatic reactions up to 10 weeks post RF treat-ment. Fractional RF treatment generated a RF ther-mal zone (RFTZ) pattern in the reticular dermis that consisted of zones of denatured collagen separated by zones of spared dermis. RFTZs were observed through day 28 post-treatment, but were replaced by new dermal tissue by 10 weeks. Reticular dermal vol-ume, cellularity, hyaluronic acid, and elastin content increased. They concluded that a vigorous wound-healing response is initiated post fractional RF treat-ment, with progressive increase in inflammatory cell infiltration from day 2 through 10 weeks. An active dermal remodeling process leads to complete replace-ment of RFTZs with new collagen by 10 weeks post-treatment. Furthermore, they demonstrated evidence of profound neoelastogenesis following RF treatment
of human skin, indicating the potential of this tech-nology to provide a reliable treatment option for skin laxity and/or rhytides.
Following this initial study, Hantash et al. (20) conducted a pilot clinical study using the same frac-tional RF microneedles device. A range of pulse durations between 1 and 25 seconds, and lesion tem-peratures between 60o and 80oC were tested in vivo on 15 human subjects. Thermal effects were assessed histologically using specific staining. Treatment effects and adverse events were also monitored clin-ically. Histological staining revealed the presence of zones of denatured collagen within the reticular dermis. Lesions were generated at preselected tem-peratures between 60 and 80oC. Fractional lesions separated by zones of spared as well as contiguous lesion patterns were demonstrated. No major adverse events were observed.
Alexiades-Armenakas et al. (21) performed a blinded, randomized, quantitative grading compari-son of microneedles and surgical facelift to treat skin laxity using the Primaeva system. Fifteen patients with facial skin laxity enrolled in the trial and completed treatment and follow-up. Baseline and follow-up dig-ital photographs of patients undergoing microneedle treatments were randomly mixed with 6 sets of base-line and follow-up images of patients undergoing sur-gical facelift with equivalent baseline facial laxity grades. Five independent blinded evaluators graded randomized baseline and 3- to 6-month follow-up photographs using a 4-point laxity grading scale. Quantitative changes in laxity grades were calculated and compared statistically for microneedle treatments versus surgical facelifts. Patient satisfaction and adverse events were also evaluated.
Results demonstrated statistically significant improvement in facial laxity, with a mean grade improvement of 1.20 for patients in the surgical face-lift group and of 0.44 for microneedles-treated patients, on a 4-point laxity grading scale (P 0.001). The mean laxity improvement, following a single microneedles treatment, was 37% than that of the surgical facelift. Patient satisfaction was high (dissat-isfied, 0%; neutral, 7%; satisfied, 60%; and very satis-fied, 33%). All participants in the microneedles treatment group experienced transient erythema, mild edema, and mild to moderate purpura that resolved in 5–10 days, and they returned to normal activities within 24 hours. There were no adverse events or complications in the microneedles group. All patients in the surgical facelift group experienced scarring at surgical margins, erythema, edema, and ecchymosis, and they returned to normal activities on suture removal at 7–10 days. The authors concluded that minimally invasive microneedles RF treatment may provide an important non-surgical option for the treatment of facial skin laxity, without the adverse effects and complications of surgical procedures. Several other RF microneedle systems are being marketed worldwide, such as Infini by Lutronic. In a company’s “white paper” publication, Calderhead et al. (22) report on the evaluation of the safety and efficacy of Infini microneedles RF treatment of facial wrinkles, in a multicenter study on 499 patients. A variety of parameters, such as power level, exposure time, and number of passes and sessions, were used according to severity of the condition and the treated area. Needles depth ranged from 0.5 to 3 mm. The study resulted in an effective and safe skin rejuvena-tion and treatment of rhytides in all Fitzpatrick skin types examined (I–V), with no secondary hyperpig-mentation in the darker Asian skin types, including Indian patients with skin type of IV–V. The results obtained after the final treatment continued to improve over time, and patient satisfaction with the procedure was very high with excellent compliance. The first microneedle technology from Pollogen Ltd. is based on fractional Hybrid Energy™ (HE), a combination of RF and galvanic energy. Boisnic and Branchet demonstrated, in a recently published arti-cle (23), the morphological and histological effects of the HE technology on epidermal and dermal skin layers, using ex vivo human skin model. Human skin samples were collected, and treated ex vivo using the HE applicator. The effect of this treatment was eval-uated by skin histology and quantitative analysis using assays of collagen fibers, elastin, and glycosami-noglycans (GAGs) dosage, reflecting hyaluronic acid content, in addition to epidermal mitotic index eval-uation. Histology results demonstrated immediate- and long-term HE effects on both epidermal and dermal skin layers with a direct correlation between the treatment parameters and treatment effect. Bio-chemical assays demonstrated a significant increase in the epidermal mitotic index, significant dermal col-lagen remodeling, and significant increase in both epidermal and dermal GAGs, indicating an increase in hyaluronic acid. It was concluded that exposure to the HE technology was shown to affect significant collagen remodeling and a significant increase in mid- to deep dermis GAGs, in addition to epidermal mitotic index, resulting in a long-term structural and biochemical dermal and epidermal improvement in treated skin samples.
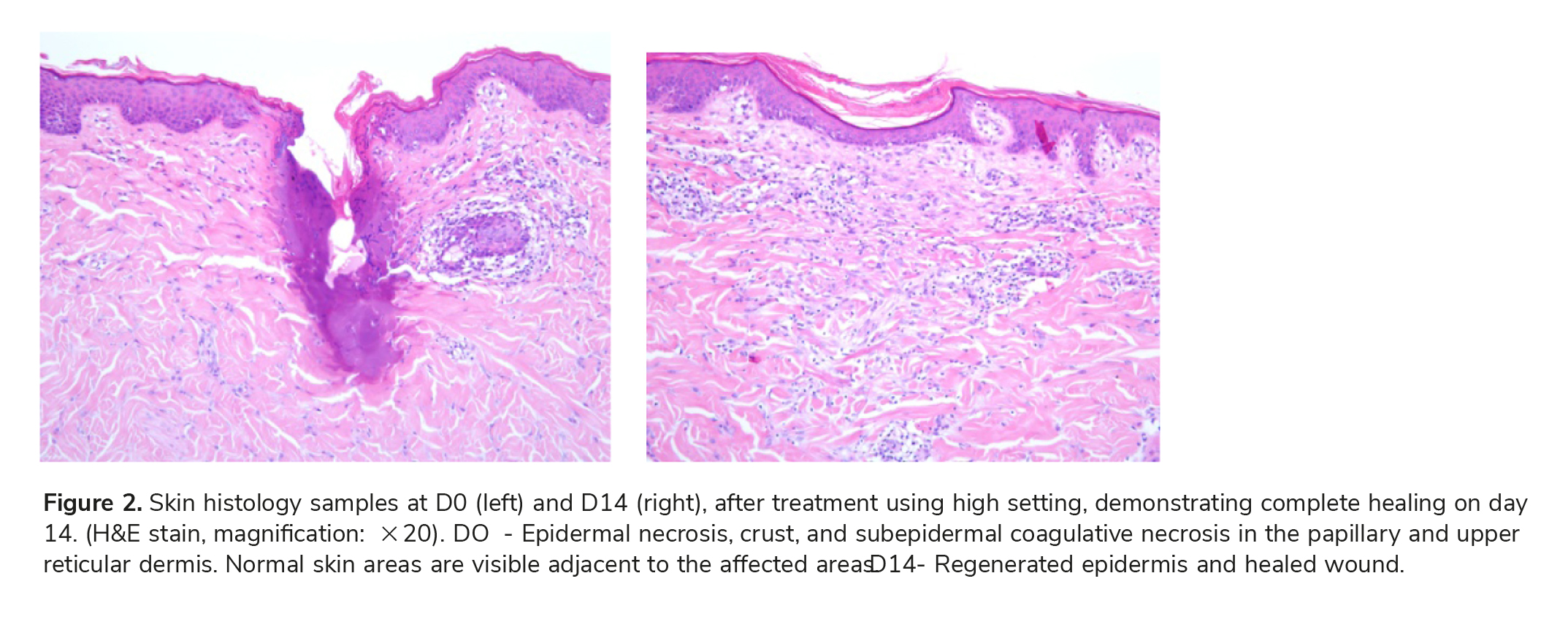
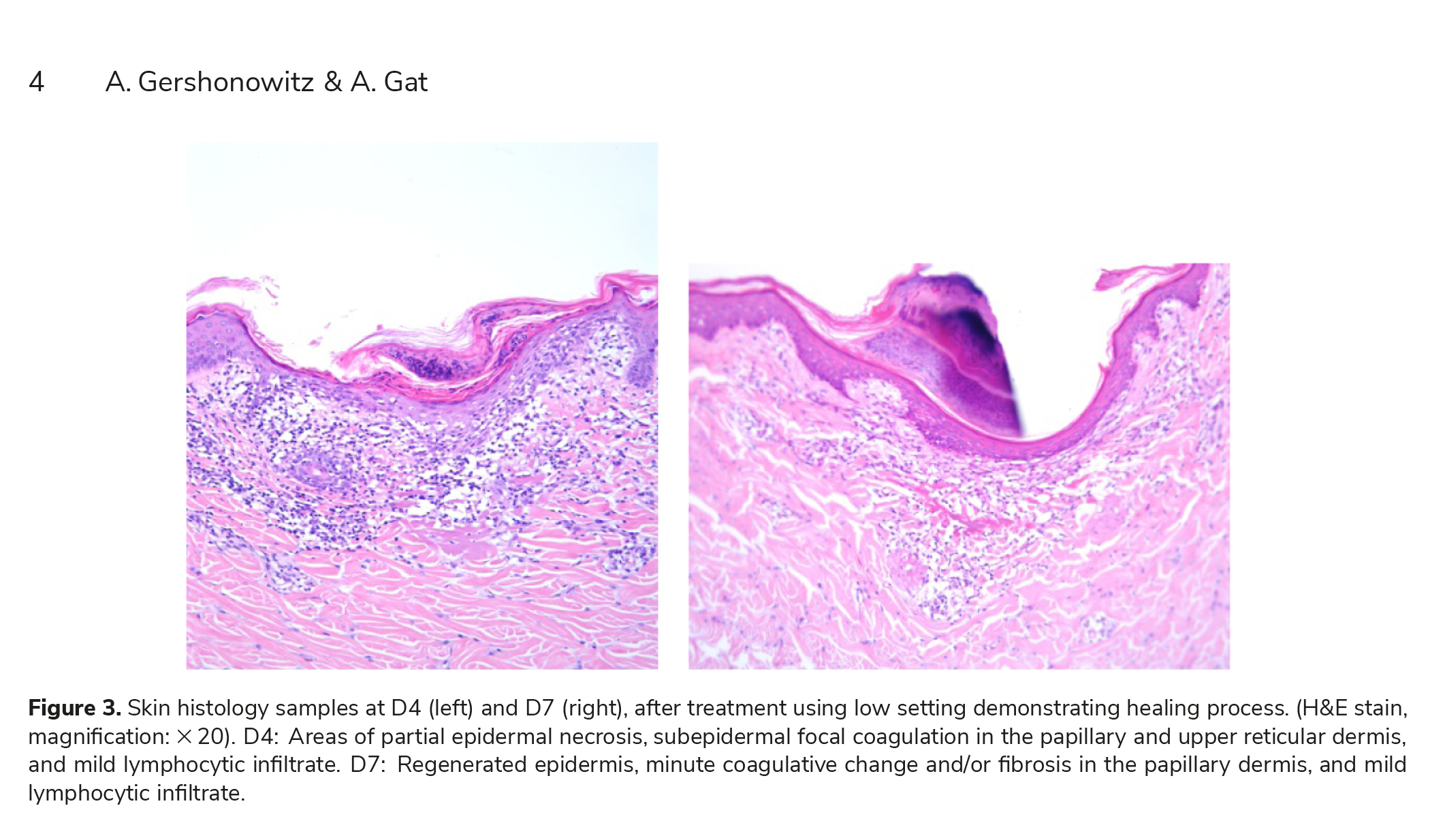
Pollogen VoluDerm and Pollogen HE technologies share a tip design and the smart RF-assisted skin pen-etration characteristics, enabling an optimal dermal impact via a bloodless, painless fractional treatment. The current histological evaluation supports the safety and efficacy of the VoluDerm RF microneedles technology. Results demonstrate optimized skin pen-etration and stimulation of natural wound-healing process. The histological results of this study demon-strate immediate (Day 0) epidermal and dermal effects following a VoluDerm treatment and a prog-ress in healing process, as a function of time follow-ing treatment (days 4 and 7). The VoluDerm treatment triggers the body’s natural healing response including blood vessels-related reaction, such as lym-phocytes infiltrate that was visible in histological samples during healing process. Complete healing was demonstrated within the 14-day period for all energy levels.
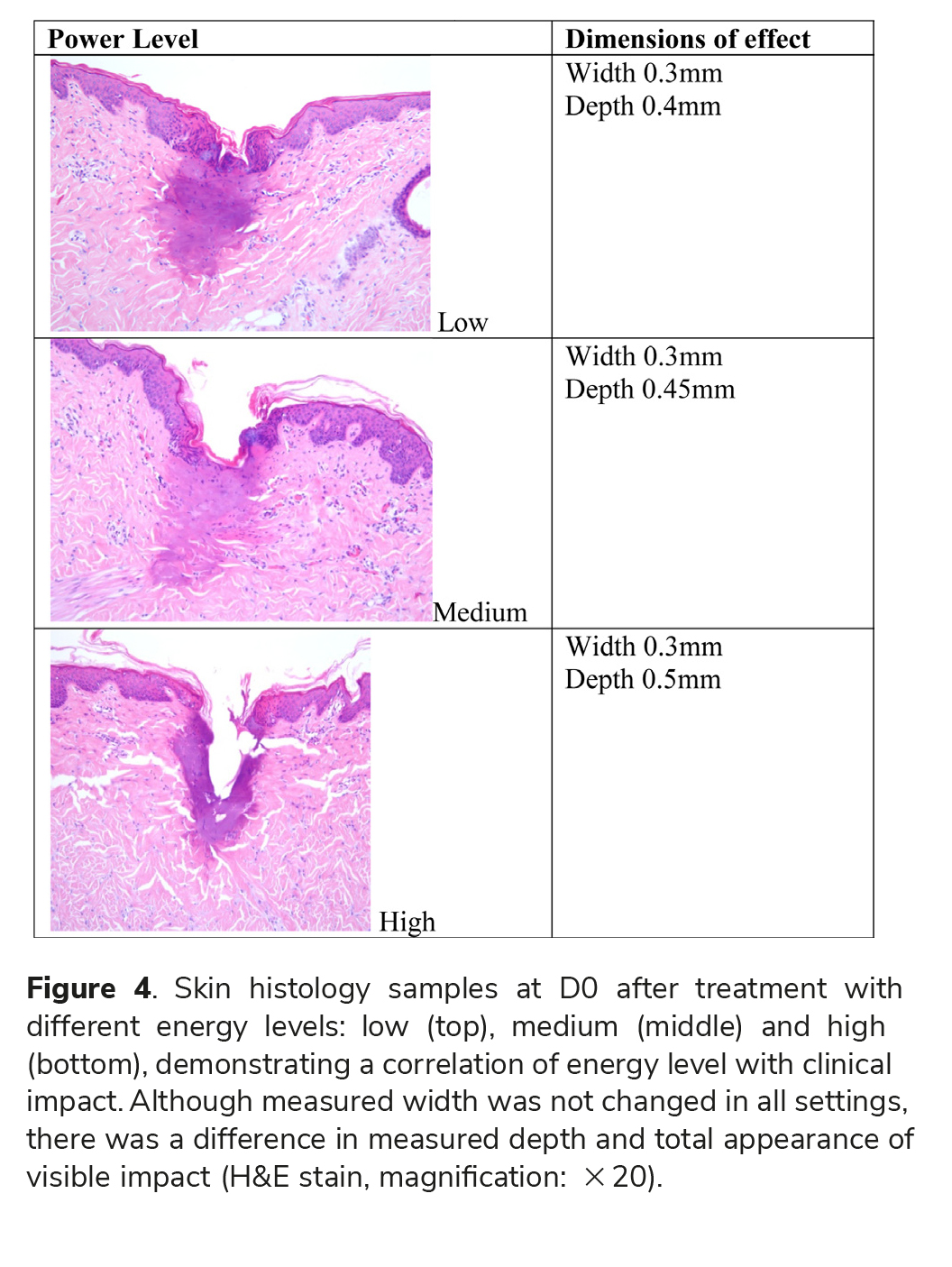
Extent of effect was correlated with the energy level. Namely, using high level resulted in the highest impact.
The results of this current study clearly indicate that treatment using the VoluDerm Applicator, with both gen 12 and gen 36 tips, is safe and effective. A complete regeneration of the epidermis and dermis with subepidermal healing was demonstrated after 14 days for all energy levels.
All treated areas, with coagulation changes or after healing, were surrounded by normal healthy tis-sue. This result indicates that the treatment induced the expected fractional pattern in treatment site, sur-rounded by normal healthy tissue.
The major impact of VoluDerm is on the dermal layer, thus treatment is associated with less to no downtime, less patient discomfort, and less predis-position to side effects compared with no-needles fractional technologies or other microneedles tech-nologies in which penetration mechanism is mechan-ical. With VoluDerm, the visible impact on the epidermal layer is relatively minor and less promi-nent compared with these technologies, although the desired micro-resurfacing effect is achieved. Due to the RF-assisted smooth penetration of the VoluDerm microneedles into the skin and to the unique tip elec-trodes design, treatments result in effective and safe treatment coverage and clinical outcome, thus there is no need for longer needles to achieve the desired clinical effect.
Declaration of interest: The authors report no declarations of interest. The authors alone are respon-sible for the content and writing of the paper. The study was funded by Pollogen Ltd.
References
1. Gold MH. Tissue tightening: a hot topic utilizing deep dermal heating. J Drugs Dermatol. 2007;6:1238–1242.
2. Alster TS, Lupton JR. Nonablative cutaneous remodeling using radiofrequency devices. Clin Dermatol. 2007;25: 487–491.
3. Sadick N, Sorhaindo L. The radiofrequency frontier: a review of radiofrequency and combined radiofrequency pulsed light technology in aesthetic medicine. Facial Plast Surg. 2005; 21:131–138.
4. Goldman P, Fitzpatrick E. Cutaneous laser surgery – The art and science of selective photothermolysis. St. Louis, MO: Mosby. 1999. p. 377.
5. Kaplan H, Gat A. Clinical and histopathological results following TriPollar radiofrequency skin treatments. J Cosmet Laser Ther. 2009;11:78–84.
6. Boisnic S, Branchet MC. Ex vivo human skin evaluation of localized fat reduction and anti-aging effect by TriPollar radio frequency treatments. J Cosmet Laser Ther. 2010;12: 25–31.
7. Levenberg A. Clinical experience with a TriPollar radiofre- quency system for facial and body aesthetic treatments. Eur J Dermatol. 2010;20:615–619. Epub 2010 Jul 13.
8. Shapiro SD, Eros Y, Abrahami Y, Leviav A. Evaluation of safety and efficacy of the TriPollar technology for treatment of wrinkles Publication. Las Surg Med. 29 Jun 2012.
9. Spadoni D, Cain CL. Facial resurfacing. Using the carbon dioxide laser. AORN J. 1989;50:1007, 1009–1013.
10. Weinstein C. Ultrapulse carbon dioxide laser removal of periocular wrinkles in association with laser blepharoplasty. J Clin Laser Med Surg. 1994;12:205–209.
11. Waldorf HA, Kauvar AN, Geronemus RG. Skin resurfacing of fine to deep rhytides using a char-free carbon dioxide laser in 47 patients. Dermatol Surg. 1995;21:940–946.
12. Teikemeier G, Goldberg DJ. Skin resurfacing with the erbium:YAG laser. Dermatol Surg. 1997;23:685–687.
13. Manstein D, Herron GS, Sink RK, Tanner H, Anderson RR. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers Surg Med. 2004;34:426–438.
14. Laubach HJ, Tannous Z, Anderson RR, Manstein D. Skin responses to fractional photothermolysis. Lasers Surg Med. 2006;38:142–149.
15. Tierney EP, Eisen RF, Hanke CW. Fractionated CO2 laser skin rejuvenation. Dermatol Ther. 2011;24:41–53.
16. Hruza G, Taub AF, Collier SL, Mulholland SR. Skin rejuvenation and wrinkle reduction using a fractional radiof-requency system. J Drugs Dermatol. 2009;8:259–265.
17. Levenberg A, Gat A, Boisnic S, Branchet MC. Treatment of wrinkles and acne scars using the TriFractional, a novel fractional radiofrequency technology – Clinical and histo-logical results. J Cosmet Dermatol Sci Appl. 2012:117–125.
18. Gold MH, Adelglass J. Evaluation of safety and efficacy of the TriFractional RF technology for treatment of facial wrinkles. J Cosmet Laser Therapy. 2014;16:2–7.
19. Hantash BM, Ubeid AA, Chang H, Kafi R, Renton B. Bipolar fractional radiofrequency treatment induces neoelas-togenesis and neocollagenesis. Lasers Surg Med. 2009;41:1–9.
20. Hantash BM, Renton B, Berkowitz RL, Stridde BC, Newman J. Pilot clinical study of a novel minimally invasive bipolar microneedle radiofrequency device. Lasers Surg Med. 2009;41:87–95.
21. Alexiades-Armenakas M, Rosenberg D, Renton B, Dover J, Arndt K. Blinded, randomized, quantitative grading com-parison of minimally invasive, fractional radiofrequency and surgical face-lift to treat skin laxity. Arch Dermatol. 2010; 146:396–405.
22. Calderhead RG, Goo BL, Lauro F, Gursoy D, Savant SS, Wronski A. The clinical efficacy and safety of microneedling fractional radiofrequency in the treatment of facial wrinkles: a multicenter study with the Infini System in 499 patients. White paper, Lutronic Corp, Goyang, South Korea.
23. Boisnic S, Branchet MC. Ex vivo study of hybrid energy tech- nology using a human skin model. Eur J Dermatol. 2014;24:46–52.
24. Martin P. Wound healing – aiming for perfect skin regenera- tion. Science. 1997;275:75–81.
25. Li WW. Angiogenesis in wound healing. A supplement to Contemporary Surgery. Nov. 2003.